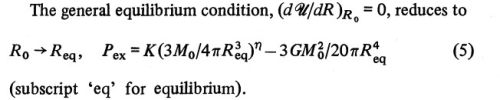SSC/Virial/PolytropesEmbedded/FirstEffortAgain/Pt2
Virial Equilibrium of Adiabatic Spheres (1st Effort)[edit]
Part I: Isolated Configurations |
Part II: Configurations Embedded in an External Medium |
Nonrotating Adiabatic Configuration Embedded in an External Medium[edit]
For a nonrotating configuration that is embedded in, and is influenced by the pressure of, an external medium, the statement of virial equilibrium is,
Solution Expressed in Terms of K and M (Whitworth's 1981 Relation)[edit]
This is precisely the same condition that derives from setting equation (3) to zero in Whitworth's (1981, MNRAS, 195, 967) discussion of the Global Gravitational Stability for One-dimensional Polytropes. The overlap with Whitworth's narative is clearer after introducing the algebraic expressions for the coefficients , , and , to obtain,
|
|
|
|
dividing the equation through by ,
|
|
|
|
|
|
|
|
and inserting expressions for the parameter normalizations as defined in our accompanying introductory discussion to obtain,
|
|
|
|
|
|
|
|
If the structural form factors are set equal to unity, this exactly matches equation (5) of Whitworth, which reads:
Notice that, when , this expression reduces to the solution we obtained for an isolated polytrope, expressed in terms of and (see the left-hand column of our table titled "Two Points of View").
Solution Expressed in Terms of M and Central Pressure[edit]
Beginning again with the relevant statement of virial equilibirum, namely,
but adopting the alternate expression for the coefficient, , given above, that is,
we can write,
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Again notice that, when , this expression reduces to the solution we obtained for an isolated polytrope, but this time expressed in terms of and (see the right-hand column of our table titled "Two Points of View").
Contrast with Detailed Force-Balanced Solution[edit]
As has just been demonstrated, the virial theorem provides a mathematical expression that allows us to relate the equilibrium radius of a configuration to the applied external pressure, once the configuration's mass and either its specific entropy or central pressure are specified. In contrast to this, as has been discussed in detail in another chapter, Horedt (1970), Whitworth (1981) and Stahler (1983) have each derived separate analytic expressions for and — given in terms of the Lane-Emden function, , and its radial derivative — without demonstrating how the equilibrium radius and external pressure directly relate to one another. That is to say, solution of the detailed force-balanced equations provides a pair of equilibrium expressions that are parametrically related to one another through the Lane-Emden function. For example — see our related discussion for more details — Horedt derives the following set of parametric equations relating the configuration's dimensionless radius, , to a specified dimensionless bounding pressure, :
|
|
|
|
|
|
|
|
where,
|
|
|
|
|
|
|
|
It is important to appreciate that, in the expressions for and , the tilde indicates that the Lane-Emden function and its derivative are to be evaluated, not at the radial coordinate, , that is traditionally associated with the "first zero" of the Lane-Emden function and therefore with the surface of the isolated polytrope, but at the radial coordinate, , where the internal pressure of the isolated polytrope equals and at which the embedded polytrope is to be truncated. The coordinate, , therefore identifies the surface of the embedded — or, pressure-truncated — polytrope. We also have taken the liberty of attaching the subscript "limit" to in both defining relations because it is clear that Hoerdt intended for the normalization mass to be the mass of the pressure-truncated object, not the mass of the associated isolated (and untruncated) polytrope. In anticipation of further derivations, below, we note here the ratio of Hoerdt's normalization parameters to ours, assuming :
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Next, we demonstrate that this pair of parametric relations satisfies the virial theorem and, in so doing, demonstrate how and may be directly related to each other. Given that Hoerdt's chosen normalization radius and normalization pressure are defined in terms of and , we begin with the virial theorem derived above in terms of and , setting .
|
|
|
|
After setting , a bit of algebraic manipulation shows that the first term on the right-hand side of the virial equilibrium expression becomes,
|
|
|
|
while the second term on the right-hand side becomes,
|
|
|
|
But, using Horedt's expression for , the left-hand side of the virial equilibrium equation becomes,
|
|
|
|
Hence, the statement of virial equilibrium is,
|
|
|
|
|
|
|
|
|
|
|
|
or, multiplying through by and rearranging terms,
|
Now, Hoerdt has given analytic expressions for and in terms of the Lane-Emden function and its first derivative. The question is, what should the expressions for our structural form factors be in order for this virial expression to hold true for all pressure-truncated polytropic structures? As has been summarized above, in the case of an isolated polytrope, whose surface is located at and whose global properties are defined by evaluation of the Lane-Emden function at , we know that (see the above summary),
|
Structural Form Factors for Isolated Polytropes | |||||||||
|---|---|---|---|---|---|---|---|---|---|
|
These same expressions may or may not work for pressure-truncated polytropes, even if the evaluation radius is shifted from to . Let's see …
Inserting Hoerdt's expressions for and into the viral equilibrium expression, we have,
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Assuming that the structural form factor, , has the same functional expression as in the case of isolated polytropes (but evaluated at instead of at ), the virial relation further reduces to the form,
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
While this does not give us individual expressions for the form factors, and , the expression derived for the ratio of the form factors makes sense because the term that has been subtracted from in the numerator on the lefthand side, that is, , naturally goes to zero in the limit of , producing the correct expression for the ratio, , in isolated polytropes. In summary, then, we have,
|
Structural Form Factors for Pressure-Truncated Polytropes | ||||||
|---|---|---|---|---|---|---|
|
Notice that, in an effort to differentiate them from their counterparts developed earlier for "isolated" polytropes, we have affixed a tilde to each of these three form-factors, .
Renormalization[edit]
Grunt Work[edit]
Returning to the dimensionless form of the virial expression and multiplying through by , we obtain,
or, after plugging in definitions of the coefficients, , , and , and rewriting explicitly as ,
|
|
|
|
This relation can be written in a more physically concise form, as follows. First, normalize to a new pressure scale — call it — and multiply through by in order to normalizing to a new length scale,:
|
|
|
|
|
|
|
|
or,
|
|
|
|
where,
|
|
|
|
|
|
|
|
By demanding that the leading coefficients of both terms on the right-hand-side of the expression are simultaneously unity — that is, by demanding that,
|
|
|
|
and,
|
|
|
|
we obtain the expressions for and as shown in the following table.
|
Renormalization for Adiabatic (ad) Systems | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
Using these new normalizations, we arrive at the desired, concise virial equilibrium relation, namely,
|
|
|
|
or,
|
|
|
|
In Terms of Free-Energy Coefficients[edit]
Referring back to relations between our free-energy coefficients, as presented earlier, we note that,
|
|
|
|
|
|
|
|
Hence, we can write,
|
|
|
|
|
|
|
|
In Terms of Horedt's Equilibrium Parameters[edit]
For later use it is also worth developing expressions for both and that are in terms of our structural form factors and Horedt's two dimensionless functions, and . (Adopting a unified notation, we will set .)
|
|
|
|
|
|
|
|
|
|
|
|
And,
|
|
|
|
|
|
|
|
|
|
|
|
Plugging in the expressions for and , as reprinted, for example, above, along with our deduced expressions for and (in terms of ), these two relations become:
|
|
|
|
|
|
|
|
|
|
|
|
and,
|
|
|
|
|
|
|
|
|
|
|
|
Summary[edit]
If we define,
|
|
|
and, |
|
|
|
|
in which case the relationship between and for pressure-truncated polytropes can be rewritten as,
|
|
|
|
In addition, the expressions for the dimensionless equilibrium radius and the dimensionless external pressure, as just derived, may be written as, respectively,
|
|
|
and, |
|
|
|
|
Using these expressions, it is easy to demonstrate that the virial equilibrium relation is satisfied, namely,
|
|
|
|
P-V Diagram[edit]
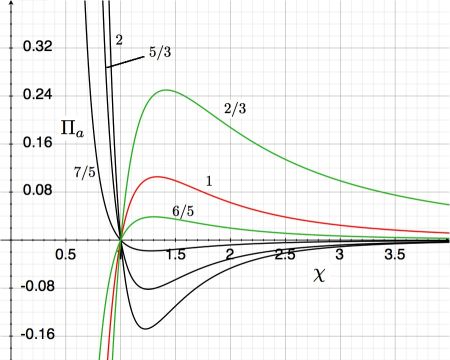
For an arbitrary value of the adiabatic exponent, , it isn't possible to invert this virial relation to obtain an analytic expression for as a function of . But, as written, the virial relation dictates the behavior of as a function of . Figure 4 displays this behavior for a number of different values of .
For physically relevant solutions, both and must be nonnegative. Hence, as is illustrated by the curves in Figure 4, the physically allowable range of equilibrium radii is,
Each of the curves drawn in Figure 4 exhibits an extremum. In each case this extremum occurs at a configuration radius, , given by,
that is, where,
For each value of , the corresponding dimensionless pressure is,
In terms of the polytropic index, the equivalent limiting expressions are,
|
|
and |
|
(In a separate, related discussion of the free-energy function, we demonstrate that this "extremum" also serves as a dividing line between dynamically stable and unstable models along a given curve.)
In examining the group of plotted curves, notice that, for , an equilibrium configuration with a positive radius can be constructed for all physically realistic — that is, for all positive — values of . Also, consistent with the behavior of the curves shown in Figure 4, the extremum arises in the regime of physically relevant — i.e., positive — pressures only for values of ; and in each case it represents a maximum limiting pressure.

Material that appears after this point in our presentation is under development and therefore
may contain incorrect mathematical equations and/or physical misinterpretations.
| Go Home |
Maximum Mass[edit]
n = 5 Polytropic[edit]
When — which corresponds to an polytropic configuration — we obtain,
which corresponds to a maximum mass for pressure-bounded polytropic configurations of,
This result can be compared to other determinations of the Bonnor-Ebert mass limit.
See Also[edit]

|
|---|
|
Appendices: | VisTrailsEquations | VisTrailsVariables | References | Ramblings | VisTrailsImages | myphys.lsu | ADS | |